The US Food and Drug Administration is warning that dangerous counterfeit versions of Botox have been identified in multiple states, putting the safety of consumers at risk.
Some serious symptoms – including blurred vision, difficulty swallowing, shortness of breath, difficulty lifting one’s head and even hospitalizations – have been linked to the use of counterfeit Botox, the FDA said on Tuesday.
“These symptoms are similar to those seen when botulinum toxin spreads to other parts of the body,” according to the FDA announcement.
The agency announced that it is working closely with the US Centers for Disease Control and Prevention, state health departments and manufacturers to investigate and identify these counterfeit products and remove them from the market.
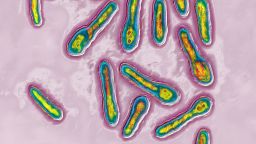
Botulinus, Clostridium botulinum, This bacterium secretes a toxin, Botox, which inhibits the neurons responsible for muscle contraction. Image produced using optical microscopy X 2000. (Photo by: BSIP/Universal Images Group via Getty Images) BSIP/Universal Images Group/Getty Images
As of Friday, a total of 19 women from nine states – Colorado, Florida, Illinois, Kentucky, Nebraska, New Jersey, New York, Tennessee, and Washington – reported “harmful reactions” after receiving Botox injections from unlicensed or untrained individuals or in places that were not health care settings, such as homes or spas, according to the CDC. The patients were between 25 and 59 years old.
Nine of the women were hospitalized and four were treated with botulism antitoxin medication due to concerns that the botulinum toxin could have spread beyond the injection site. Five women were tested for the illness botulism. Their results were negative.
The incidents of counterfeit Botox being administered to consumers appear to be connected to products that have been purchased from unlicensed sources and then administered by either unlicensed or licensed providers.
The FDA warns health care professionals that purchasing and administering counterfeit products puts patients’ health at risk and all providers should check products for any signs of counterfeiting before using them.
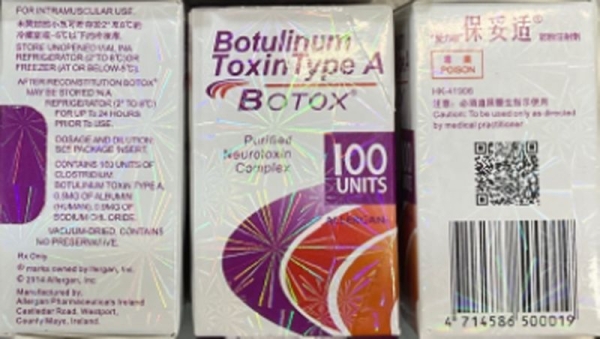
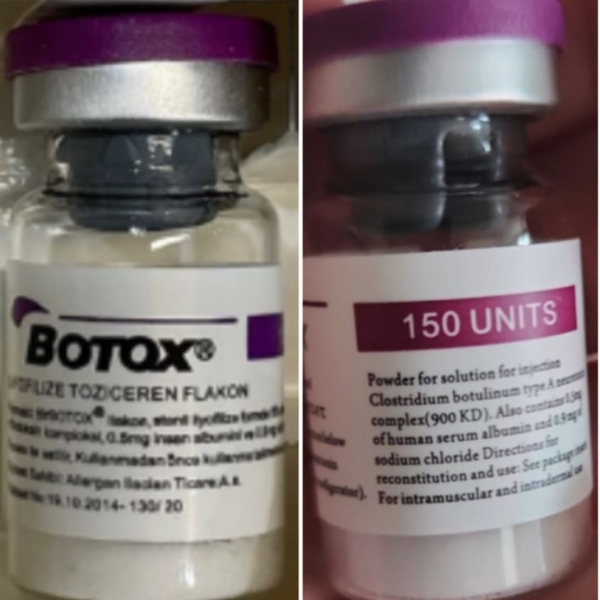
The counterfeit Botox products may be identified by the lot number C3709C3 found on the outer carton and vial, the FDA said.
The outer carton also may display the active ingredient as “Botulinum Toxin Type A” instead of “OnabotulinumtoxinA.” Also, the carton and vial may indicate 150-unit doses, which is not a unit made by the companies AbbVie or Allergan, the FDA said, and the outer carton may contain language that is not English.
Counterfeit botox has been found in several states. Here’s what consumers should know. FDA
What questions you should ask
The growing spread of counterfeit Botox has horrified Dr. Lavanya Krishnan, a board-certified dermatologist in San Francisco who founded the practice Arya Derm and has been following the FDA and CDC warnings closely.
“I think it’s horrific and honestly really sad to think that someone is going in for what should be a very simple, straightforward aesthetic injection and walking out with symptoms that are classic for botulism,” Krishnan said.
In the United States, there are five different types of FDA-approved neurotoxins used for cosmetic purposes, according to the American Board of Cosmetic Surgery: Botox under the company Allergan; Dysport under the company Galderma; Jeuveau under the company Evolus; Daxxify under Revance Therapeutics and Xeomin under Merz Aesthetics. Providers purchase these products directly from the company manufacturers to ensure that they are getting the real FDA-approved product, Krishnan said.
“So, the best way to find a licensed provider is to go to the company websites for these products – say you wanted Botox, you would go to the company Allergan’s website, and Allergan actually should have a list of licensed providers who they are selling their product to,” Krishnan said, adding that it also can be helpful for consumers to ask their providers where they are getting their product from.

Shipments of unregulated fillers from Bulgaria, Spain, China and Korea were being sent to Oregon, South Carolina, New York and other US locations, authorities said. US Customs and Border Protection
“Counterfeit products are often quoted at much lower prices, and that should be a tip-off to ask more questions about where that product was purchased from, and patients have a right to know,” Krishnan said.
“Sometimes when the price is too good to be true, that should be a tip-off,” she said. “Brand name Botox typically costs a certain amount. Prices can vary a little bit from clinic to clinic, based on the practitioner and based on the location of the clinic, but if the price is way cheaper, I think that sometimes that should be a flag.”
If a provider is not licensed then the manufacturer won’t sell them their product, said plastic surgeon Dr. Jimmy Sung, medical director of Tribeca Aesthetics in New York City, who has treated patients with adverse reactions from previous cosmetic services.
“First of all, if you’re not licensed, you can’t get the drug legitimately. The manufacturer will not sell it to you,” Sung said.
Most often, when counterfeit Botox is sold, “the injector knows they’re purchasing something counterfeit,” Sung said. “And the counterfeiter knows that they’re selling to somebody who’s most likely not a board-certified plastic surgeon or a board-certified dermatologist.”
The symptoms to watch for
Botox as a product contains the neurotoxic protein botulinum toxin, which, when used in small doses, can treat various medical or cosmetic concerns, such as severe underarm sweating, chronic migraines, overactive bladder or temporarily smoothing facial wrinkles.
“Botox and its peer brands provide a dilute, precise, and consistent amount of botulinum toxin, whereas knock-off unregulated products are not reliably manufactured or subject to the same quality assurance, which can lead to over or underdosing,” said Dr. Michael Cameron, president and founder of Cameron Dermatology and assistant clinical professor of dermatology at Mount Sinai Health System in New York.
When in the body at high levels, the toxin botulinum can cause the rare but serious illness botulism, or symptoms similar to those from the illness.

A woman’s bare shoulders Bernd Vogel/Stone RF/Getty Images
“Signs and symptoms of botulism include muscle weakness, vision changes, slurred speech, difficulty moving your eyes. An emergency would be if you start to have difficulty breathing,” Cameron said.
“I personally recommend if you’re getting Botox for your wrinkles, you go to a board-certified dermatologist. If you’re getting Botox for your migraines, you go to a neurologist. If you’re getting Botox for your bladder issues, you go to your urologist,” Cameron said. “If you feel like there’s any risk or the pricing doesn’t really make sense, then you could just ask to actually see the vial and inspect the vial of Botox yourself.”
Sung said that counterfeit Botox could be made in a cheaper way that makes it more potent, causing serious reactions, or it could be made in a way that makes it more easily spreadable once it’s injected under the skin, raising the risks of it traveling to other parts of the face or body where it was not initially intended for.
“The factories that make fake Botox usually don’t have the tightest quality control. They could have a bad batch that’s more potent, that spreads out a little more than the next batch,” Sung said. “It’s no different than getting a bad batch of fentanyl or heroin on the street.”
Counterfeit toxins, used for cosmetic purposes, seem to have been circling on the market for some time, Sung said.
“I’m actually surprised that it took like, almost 20 years for the FDA to actually issue something. Although I’ve never seen it, I’m aware that this is going on,” he said. “I’m aware because patients tell me.”
How to find the right provider
If a provider is hesitant or deceptive in sharing details about their training, their credentials, or where they purchased product from, that behavior should be a “massive red flag” and patients should not be afraid to ask questions or reconsider whether they are at the right place to receive these types of treatments, Dr. Steven Williams, president of the American Society of Plastic Surgeons, said in an email Tuesday.
“Providers that won’t show you the original bottle or packaging that the Botox came in should be viewed with some suspicion,” Williams said. He added that rules around injecting medications like Botox can vary somewhat from state to state, but in most states, nurses, physician assistants and physicians can inject Botox, but in some places, it is required for nurses to be supervised by physicians when administering the injections.
“Patients should be suspicious if they are offered Botox injections in hotel rooms or at home. Generally, the most qualified physician, physician assistant, or nurse will be working at a plastic surgeon or dermatology office,” Williams said. “These providers are not only expert injectors of these types of medications but are well prepared to manage potential complications and advise on the risks and benefits of these treatments.”
Get CNN Health’s weekly newsletter
Sign up here to get The Results Are In with Dr. Sanjay Gupta every Tuesday from the CNN Health team.
Other steps that patients can take to find the right provider including checking patient reviews for the provider’s practice and making sure there aren’t any that describe concerning side effects from the Botox, such as slurred speech, difficulty breathing or double vision, Dr. Cynthia Elliott, owner and primary practitioner of Skinspirations in Florida, said in an email Tuesday.
“Read the reviews that aren’t 5 stars to see what patients were not happy about and if there are a lot describing bad outcomes that sound legitimate, skip that practice because there are plenty of better ones,” Elliott said.
Also, “check the before and after photos used on the provider’s website to confirm that they are their own patients’ photos. There’s often a watermark in the photo so that it can’t be used to show results on another provider’s website. Be wary if they only have before and after photos published by the manufacturer,” she said, adding, “Make an appointment for a consultation with the provider to get a feel for their knowledge before committing to a treatment. Some practices only allow you to meet with what is essentially a salesperson during your consultation, which is a red flag.”

